 X-ray diffraction data were collected at Diamond Light Source (Oxfordshire, U.K.) at the I04-1 beamline at wavelength 0.91587 Å. Crystals were cryoprotected using a 30% solution of ethylene glycol and then flash-cooled at 100 K. Diffraction quality crystals were grown in the following conditions: 0.02 M sodium/potassium phosphate, 20% PEG 3350 (NYE_S2 TCR–pHLA) and 0.2 M ammonium sulfate, 15% PEG 8000, and 0.1 M Tris (pH 7.5) (NYE_S3 TCR–pHLA). Plates were maintained at 20☌ in a Rock Imager 1000 (Formulatrix) storage system. Diffraction images were indexed, integrated, scaled, and merged using DIALS ( 39) and Aimless ( 40) through the xia2 processing suite ( 41).įor NYE_S2 and NYE_S3 TCR–pHLA complexes, crystallization trials using 150 nl protein solution plus 150 nl reservoir solution in sitting-drop vapor diffusion format were set up in two-well MRC Crystallization plates using a Crystal Gryphon (Art Robbins) robot. X-ray diffraction data were collected at Diamond Light Source on beamline I02. 
Crystals were cryoprotected by addition of 15% glycerol directly to the drop and then flash-cooled at 100 K. Larger crystals were grown by cross-seeding into a grid of 10–25% PEG 4000, 0.1 M sodium citrate (pH 5–6), 0.2 M ammonium sulfate using a Seed Bead–generated seed solution and by increasing the drop size to 1 μl protein plus 1 μl well solution. Initial crystals grew in the PEGs II Crystallization Screen (QIAGEN), condition F6. Crystallization trials, using 100 nl protein solution plus 100 nl reservoir solution in sitting-drop vapor diffusion format, were set up in two-well MRC Crystallization plates using a mosquito (TTP Labtech) robot. To achieve this, TCRs typically engage a small number of exposed antigenic features or “hotspots,” whereas other peptide positions at the periphery of the TCR–pHLA interface show greater amino acid permissivity ( 6).įor NYE_S1, TCR–pHLA complex was prepared by mixing purified TCR and pHLA at equimolar ratios. TCRs must also simultaneously display remarkable specificity for distinguishing foreign Ag from the more limited HLA-specific self-peptide repertoire. Thus, TCR cross-reactivity or polyspecificity is thought to be essential for adequate recognition of the potential pathogenic repertoire. 
However, for the ∼1 × 10 7−8 unique TCRs present in the body to recognize the entire peptide repertoire, any given TCR must be capable of recognizing up to a million distinct theoretical peptides ( 4, 5). The specificity of TCRs toward a given peptide–HLA (pHLA) complex ensures appropriately targeted natural T cell responses. 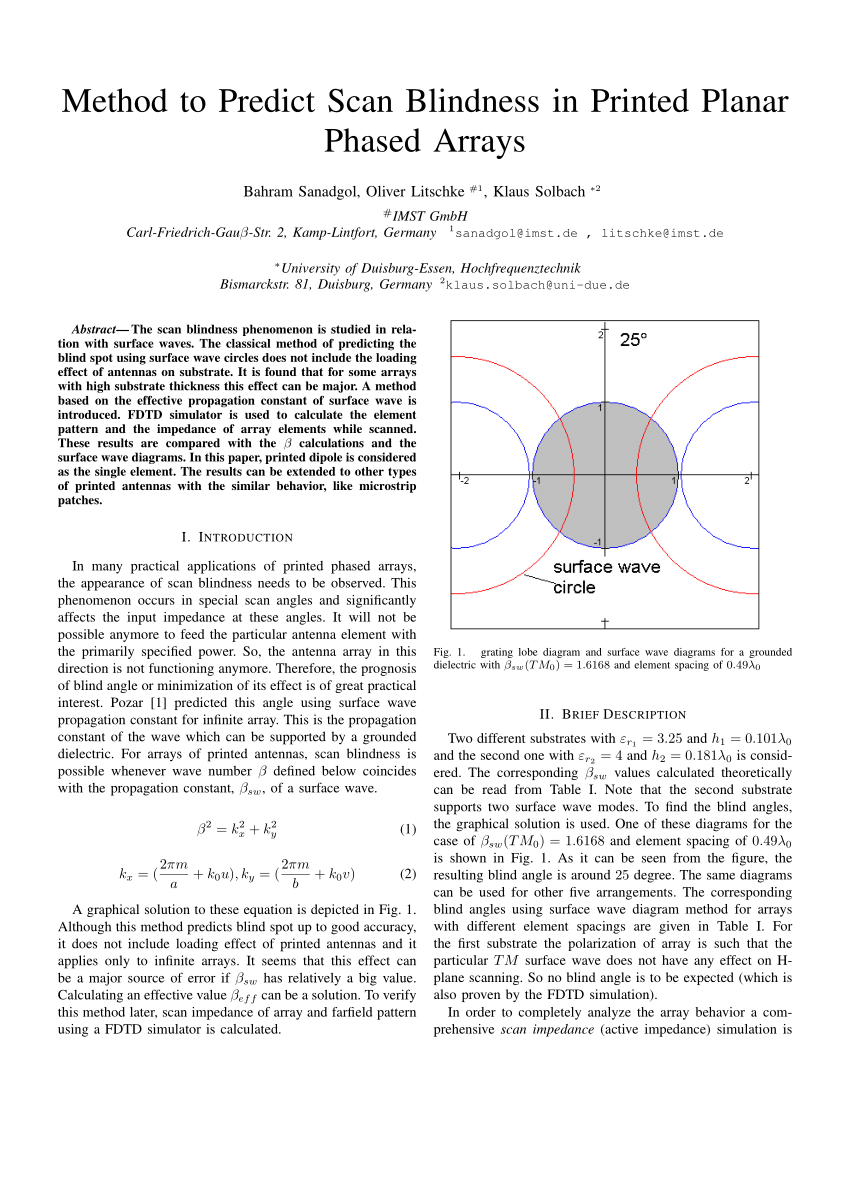
The presentation of intracellularly processed peptides by class I HLA molecules on the surface of cancer or virally infected cells enables their direct recognition and elimination by CD8 + αβ T cells ( 1– 3). These data show that TCRs specific for a cognate peptide recognize discrete peptide repertoires and reconciles how an individual’s limited TCR repertoire following negative selection in the thymus is able to recognize a vastly larger antigenic pool. The third TCR engaged a flipped peptide conformation, leading to the recognition of off-target peptides sharing little similarity with the cognate peptide. Two TCRs engaged the same central peptide feature, although were more permissive at peripheral peptide positions and, accordingly, possessed partially overlapping peptide specificity profiles. We determined TCR–peptide–HLA crystal structures and, using a single-chain peptide–HLA phage library, we generated peptide specificity profiles for three newly identified human TCRs specific for the cancer testis Ag NY-ESO-1 157–165–HLA-A2. The molecular rules driving TCR cross-reactivity are poorly understood and, consequently, it is unclear the extent to which TCRs targeting the same Ag recognize the same off-target peptides. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
